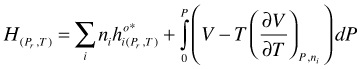H as function of P and T. Integration of gives:
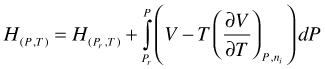
(101)
For an ideal gas:
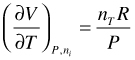
(102)
As shown in (103) the integral in (101) is zero and H for an ideal gas is not a function of P
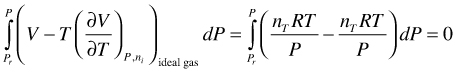
(103)
Expanding the lower integration boundary we get:
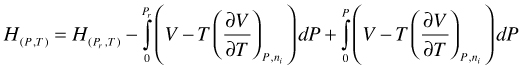
(104)
Combination of the first two terms of the right hand side of (104) yields H for a hypothetical ideal gas. In addition, for an ideal gas mixture there is no excess H
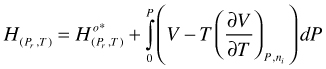
(105)